Abstract
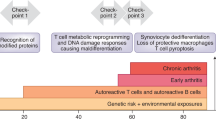
In rheumatoid arthritis, juvenile idiopathic arthritis and other forms of inflammatory arthritis, the immune system targets certain joints but not others. The pattern of joints affected varies by disease and by individual, with flares most commonly involving joints that were previously inflamed. This phenomenon, termed joint-specific memory, is difficult to explain by systemic immunity alone. Mechanisms of joint-specific memory include the involvement of synovial resident memory T cells that remain in the joint during remission and initiate localized disease recurrence. In addition, arthritis-induced durable changes in synovial fibroblasts and macrophages can amplify inflammation in a site-specific manner. Together with ongoing systemic processes that promote extension of arthritis to new joints, these local factors set the stage for a stepwise progression in disease severity, a paradigm for arthritis chronicity that we term the joint accumulation model. Although durable drug-free remission through early treatment remains elusive for most forms of arthritis, the joint accumulation paradigm defines new therapeutic targets, emphasizes the importance of sustained treatment to prevent disease extension to new joints, and identifies a rolling window of opportunity for altering the natural history of arthritis that extends well beyond the initiation phase of disease.
Key points
-
In rheumatoid arthritis and juvenile idiopathic arthritis, joints previously affected are more likely to flare than joints previously spared, a phenomenon termed joint-specific memory.
-
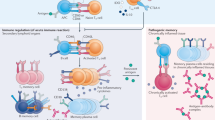
One mechanism underlying joint-specific memory is the accumulation of synovial resident memory T (TRM) cells, long-lived resident lymphocytes that remain during remission and promote recurrent disease.
-
Synovial TRM cells are predominantly CD8+ T cells and exhibit a restricted TCR repertoire; in animal models, TRM cells trigged by specific antigens induce arthritis flare by releasing chemokines, including CCL5.
-
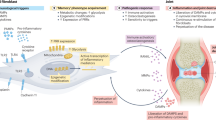
Mechanisms that predispose individual joints to greater disease activity include inflammation-induced fibroblast priming and an increase in the abundance of pro-inflammatory macrophages compared with that of regulatory macrophages.
-
Recognition that both systemic factors and local factors drive arthritis suggests a new paradigm of chronicity, the joint accumulation model, implying a ‘rolling window of opportunity’ for therapy, even in established arthritis.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Chang, M. H. & Nigrovic, P. A. Antibody-dependent and -independent mechanisms of inflammatory arthritis. JCI Insight 4, https://doi.org/10.1172/jci.insight.125278 (2019).
Gravallese, E. M. & Firestein, G. S. Rheumatoid arthritis — common origins, divergent mechanisms. N. Engl. J. Med. 388, 529–542 (2023).
Roberts, W. N., Daltroy, L. H. & Anderson, R. J. Stability of normal joint findings in persistent classic rheumatoid arthritis. Arthritis Rheum. 31, 267–271 (1988).
Chang, M. H. et al. Joint-specific memory and sustained risk for new joint accumulation in autoimmune arthritis. Arthritis Rheumatol. 74, 1851–1858 (2022).
Chang, M. H. et al. Arthritis flares mediated by tissue-resident memory T cells in the joint. Cell Rep. 37, 109902 (2021).
Heckert, S. L. et al. Joint inflammation tends to recur in the same joints during the rheumatoid arthritis disease course. Ann. Rheum. Dis. 81, 169–174 (2022).
Heckert, S. L. et al. Patterns of clinical joint inflammation in juvenile idiopathic arthritis. RMD Open 9, e002941 (2023).
Gebhardt, T., Palendira, U., Tscharke, D. C. & Bedoui, S. Tissue-resident memory T cells in tissue homeostasis, persistent infection, and cancer surveillance. Immunol. Rev. 283, 54–76 (2018).
Szabo, P. A., Miron, M. & Farber, D. L. Location, location, location: tissue resident memory T cells in mice and humans. Sci. Immunol. 4, eaas9673 (2019).
Boniface, K. et al. Vitiligo skin is imprinted with resident memory CD8 T cells expressing CXCR3. J. Invest. Dermatol. 138, 355–364 (2018).
Richmond, J. M. et al. Resident memory and recirculating memory T cells cooperate to maintain disease in a mouse model of vitiligo. J. Invest. Dermatol. 139, 769–778 (2019).
Boyman, O. et al. Spontaneous development of psoriasis in a new animal model shows an essential role for resident T cells and tumor necrosis factor-α. J. Exp. Med. 199, 731–736 (2004).
Samat, A. A. K., van der Geest, J., Vastert, S. J., van Loosdregt, J. & van Wijk, F. Tissue-resident memory T cells in chronic inflammation-local cells with systemic effects? Cells 10, 409 (2021).
Fonseca, R. et al. Developmental plasticity allows outside-in immune responses by resident memory T cells. Nat. Immunol. 21, 412–421 (2020).
Wijeyesinghe, S. et al. Expansible residence decentralizes immune homeostasis. Nature 592, 457–462 (2021).
Mueller, S. N. & Mackay, L. K. Tissue-resident memory T cells: local specialists in immune defence. Nat. Rev. Immunol. 16, 79–89 (2016).
Heeg, M. & Goldrath, A. W. Insights into phenotypic and functional CD8+ TRM heterogeneity. Immunol. Rev. 316, 8–22 (2023).
Kumar, B. V. et al. Human tissue-resident memory T cells are defined by core transcriptional and functional signatures in lymphoid and mucosal sites. Cell Rep. 20, 2921–2934 (2017).
Crowl, J. T. et al. Tissue-resident memory CD8+ T cells possess unique transcriptional, epigenetic and functional adaptations to different tissue environments. Nat. Immunol. 23, 1121–1131 (2022).
Mackay, L. K. et al. Hobit and Blimp1 instruct a universal transcriptional program of tissue residency in lymphocytes. Science 352, 459–463 (2016).
Poon, M. M. L. et al. Tissue adaptation and clonal segregation of human memory T cells in barrier sites. Nat. Immunol. 24, 309–319 (2023).
Christo, S. N. et al. Discrete tissue microenvironments instruct diversity in resident memory T cell function and plasticity. Nat. Immunol. 22, 1140–1151 (2021).
Lin, Y. H. et al. Small intestine and colon tissue-resident memory CD8+ T cells exhibit molecular heterogeneity and differential dependence on Eomes. Immunity 56, 207–223.e8 (2023).
Schenkel, J. M. et al. IL-15-independent maintenance of tissue-resident and boosted effector memory CD8 T cells. J. Immunol. 196, 3920–3926 (2016).
FitzPatrick, M. E. B. et al. Human intestinal tissue-resident memory T cells comprise transcriptionally and functionally distinct subsets. Cell Rep. 34, 108661 (2021).
Milner, J. J. et al. Heterogenous populations of tissue-resident CD8+ T cells are generated in response to infection and malignancy. Immunity 52, 808–824.e7 (2020).
Frizzell, H. et al. Organ-specific isoform selection of fatty acid-binding proteins in tissue-resident lymphocytes. Sci. Immunol. 5, https://doi.org/10.1126/sciimmunol.aay9283 (2020).
Pan, Y. et al. Survival of tissue-resident memory T cells requires exogenous lipid uptake and metabolism. Nature 543, 252–256 (2017).
Howie, D., Ten Bokum, A., Necula, A. S., Cobbold, S. P. & Waldmann, H. The role of lipid metabolism in T lymphocyte differentiation and survival. Front. Immunol. 8, 1949 (2017).
Jin, R. et al. Role of FABP5 in T cell lipid metabolism and function in the tumor microenvironment. Cancers 15, https://doi.org/10.3390/cancers15030657 (2023).
Steinert, E. M. et al. Quantifying memory CD8 T cells reveals regionalization of immunosurveillance. Cell 161, 737–749 (2015).
Jung, J. et al. Synovial fluid CD69+CD8+ T cells with tissue-resident phenotype mediate perforin-dependent citrullination in rheumatoid arthritis. Clin. Transl. Immunol. 9, e1140 (2020).
Guggino, G., Rizzo, A., Mauro, D., Macaluso, F. & Ciccia, F. Gut-derived CD8+ tissue-resident memory T cells are expanded in the peripheral blood and synovia of SpA patients. Ann. Rheum. Dis. 80, e174 (2021).
Horai, R. et al. Development of chronic inflammatory arthropathy resembling rheumatoid arthritis in interleukin 1 receptor antagonist-deficient mice. J. Exp. Med. 191, 313–320 (2000).
Zhang, F. et al. Defining inflammatory cell states in rheumatoid arthritis joint synovial tissues by integrating single-cell transcriptomics and mass cytometry. Nat. Immunol. 20, 928–942 (2019).
Jonsson, A. H. et al. Granzyme K+ CD8 T cells form a core population in inflamed human tissue. Sci. Transl. Med. 14, eabo0686 (2022).
Petrelli, A. et al. PD-1+CD8+ T cells are clonally expanding effectors in human chronic inflammation. J. Clin. Invest. 128, 4669–4681 (2018).
Vanni, A. et al. Clonally expanded PD-1-expressing T cells are enriched in synovial fluid of juvenile idiopathic arthritis patients. Eur. J. Immunol. 53, e2250162 (2023).
Maschmeyer, P. et al. Antigen-driven PD-1+ TOX+ BHLHE40+ and PD-1+ TOX+ EOMES+ T lymphocytes regulate juvenile idiopathic arthritis in situ. Eur. J. Immunol. 51, 915–929 (2021).
Steel, K. J. A. et al. Polyfunctional, proinflammatory, tissue-resident memory phenotype and function of synovial interleukin-17A+CD8+ T cells in psoriatic arthritis. Arthritis Rheumatol. 72, 435–447 (2020).
Povoleri, G. A. M. et al. Psoriatic and rheumatoid arthritis joints differ in the composition of CD8+ tissue-resident memory T cell subsets. Cell Rep. 42, 112514 (2023).
Penkava, F. et al. Single-cell sequencing reveals clonal expansions of pro-inflammatory synovial CD8 T cells expressing tissue-homing receptors in psoriatic arthritis. Nat. Commun. 11, 4767 (2020).
Qaiyum, Z., Gracey, E., Yao, Y. & Inman, R. D. Integrin and transcriptomic profiles identify a distinctive synovial CD8+ T cell subpopulation in spondyloarthritis. Ann. Rheum. Dis. 78, 1566–1575 (2019).
Sasson, S. C., Gordon, C. L., Christo, S. N., Klenerman, P. & Mackay, L. K. Local heroes or villains: tissue-resident memory T cells in human health and disease. Cell Mol. Immunol. 17, 113–122 (2020).
Arazi, A. et al. The immune cell landscape in kidneys of patients with lupus nephritis. Nat. Immunol. 20, 902–914 (2019).
Zhou, M. et al. JAK/STAT signaling controls the fate of CD8+CD103+ tissue-resident memory T cell in lupus nephritis. J. Autoimmun. 109, 102424 (2020).
Boothby, I. C. et al. Early-life inflammation primes a T helper 2 cell-fibroblast niche in skin. Nature 599, 667–672 (2021).
Nygaard, G. & Firestein, G. S. Restoring synovial homeostasis in rheumatoid arthritis by targeting fibroblast-like synoviocytes. Nat. Rev. Rheumatol. 16, 316–333 (2020).
Bottini, N. & Firestein, G. S. Duality of fibroblast-like synoviocytes in RA: passive responders and imprinted aggressors. Nat. Rev. Rheumatol. 9, 24–33 (2013).
Nakano, K., Whitaker, J. W., Boyle, D. L., Wang, W. & Firestein, G. S. DNA methylome signature in rheumatoid arthritis. Ann. Rheum. Dis. 72, 110–117 (2013).
Whitaker, J. W. et al. An imprinted rheumatoid arthritis methylome signature reflects pathogenic phenotype. Genome Med. 5, 40 (2013).
Ai, R. et al. DNA methylome signature in synoviocytes from patients with early rheumatoid arthritis compared to synoviocytes from patients with longstanding rheumatoid arthritis. Arthritis Rheumatol. 67, 1978–1980 (2015).
Crowley, T. et al. Priming in response to pro-inflammatory cytokines is a feature of adult synovial but not dermal fibroblasts. Arthritis Res. Ther. 19, 35 (2017).
Friscic, J. et al. The complement system drives local inflammatory tissue priming by metabolic reprogramming of synovial fibroblasts. Immunity 54, 1002–1021.e10 (2021).
Friscic, J. et al. Reset of inflammatory priming of joint tissue and reduction of the severity of arthritis flares by bromodomain inhibition. Arthritis Rheumatol. 75, 517–532 (2023).
Lefevre, S. et al. Synovial fibroblasts spread rheumatoid arthritis to unaffected joints. Nat. Med. 15, 1414–1420 (2009).
Orange, D. E. et al. RNA identification of PRIME cells predicting rheumatoid arthritis flares. N. Engl. J. Med. 383, 218–228 (2020).
Frank-Bertoncelj, M. et al. Epigenetically-driven anatomical diversity of synovial fibroblasts guides joint-specific fibroblast functions. Nat. Commun. 8, 14852 (2017).
Ciurea, A. et al. Joint-level responses to tofacitinib and methotrexate: a post hoc analysis of data from ORAL Start. Arthritis Res. Ther. 25, 185 (2023).
Kurowska-Stolarska, M. & Alivernini, S. Synovial tissue macrophages in joint homeostasis, rheumatoid arthritis and disease remission. Nat. Rev. Rheumatol. 18, 384–397 (2022).
Alivernini, S. et al. Distinct synovial tissue macrophage subsets regulate inflammation and remission in rheumatoid arthritis. Nat. Med. 26, 1295–1306 (2020).
Misharin, A. V. et al. Nonclassical Ly6C− monocytes drive the development of inflammatory arthritis in mice. Cell Rep. 9, 591–604 (2014).
Culemann, S. et al. Locally renewing resident synovial macrophages provide a protective barrier for the joint. Nature 572, 670–675 (2019).
Montgomery, A. B. et al. Tissue-resident, extravascular Ly6c− monocytes are critical for inflammation in the synovium. Cell Rep. 42, 112513 (2023).
Alivernini, S. et al. Synovial features of patients with rheumatoid arthritis and psoriatic arthritis in clinical and ultrasound remission differ under anti-TNF therapy: a clue to interpret different chances of relapse after clinical remission? Ann. Rheum. Dis. 76, 1228–1236 (2017).
Aegerter, H. et al. Influenza-induced monocyte-derived alveolar macrophages confer prolonged antibacterial protection. Nat. Immunol. 21, 145–157 (2020).
Guilliams, M. & Svedberg, F. R. Does tissue imprinting restrict macrophage plasticity? Nat. Immunol. 22, 118–127 (2021).
Hanlon, M. M. et al. Rheumatoid arthritis macrophages are primed for inflammation and display bioenergetic and functional alterations. Rheumatology 62, 2611–2620 (2023).
Vickovic, S. et al. Three-dimensional spatial transcriptomics uncovers cell type localizations in the human rheumatoid arthritis synovium. Commun. Biol. 5, 129 (2022).
Iijima, N. & Iwasaki, A. T cell memory. A local macrophage chemokine network sustains protective tissue-resident memory CD4 T cells. Science 346, 93–98 (2014).
Vu, T. T., Koguchi-Yoshioka, H. & Watanabe, R. Skin-resident memory T cells: pathogenesis and implication for the treatment of psoriasis. J. Clin. Med. 10, https://doi.org/10.3390/jcm10173822 (2021).
Puig, L. et al. The biological basis of disease recurrence in psoriasis: a historical perspective and current models. Br. J. Dermatol. 186, 773–781 (2022).
Matos, T. R. et al. Clinically resolved psoriatic lesions contain psoriasis-specific IL-17-producing ɑβ T cell clones. J. Clin. Invest. 127, 4031–4041 (2017).
Li, X., Jiang, M., Chen, X. & Sun, W. Etanercept alleviates psoriasis by reducing the Th17/Treg ratio and promoting M2 polarization of macrophages. Immun. Inflamm. Dis. 10, e734 (2022).
Mehta, H. et al. Differential changes in inflammatory mononuclear phagocyte and T-cell profiles within psoriatic skin during treatment with guselkumab vs. secukinumab. J. Invest. Dermatol. 141, 1707–1718.e9 (2021).
Whitley, S. K. et al. Local IL-23 is required for proliferation and retention of skin-resident memory TH17 cells. Sci. Immunol. 7, eabq3254 (2022).
Dong, C., Lin, L. & Du, J. Characteristics and sources of tissue-resident memory T cells in psoriasis relapse. Curr. Res. Immunol. 4, 100067 (2023).
Monti, S., Montecucco, C., Bugatti, S. & Caporali, R. Rheumatoid arthritis treatment: the earlier the better to prevent joint damage. RMD Open 1, e000057 (2015).
Fraenkel, L. et al. 2021 American College of Rheumatology guideline for the treatment of rheumatoid arthritis. Arthritis Care Res. 73, 924–939 (2021).
Boers, M. Understanding the window of opportunity concept in early rheumatoid arthritis. Arthritis Rheum. 48, 1771–1774 (2003).
van Nies, J. A. et al. What is the evidence for the presence of a therapeutic window of opportunity in rheumatoid arthritis? A systematic literature review. Ann. Rheum. Dis. 73, 861–870 (2014).
Burgers, L. E., Raza, K. & van der Helm-van Mil, A. H. Window of opportunity in rheumatoid arthritis — definitions and supporting evidence: from old to new perspectives. RMD Open 5, e000870 (2019).
Nigrovic, P. A. Review: Is there a window of opportunity for treatment of systemic juvenile idiopathic arthritis? Arthritis Rheumatol. 66, 1405–1413 (2014).
Nigrovic, P. A. et al. Biological classification of childhood arthritis: roadmap to a molecular nomenclature. Nat. Rev. Rheumatol. 17, 257–269 (2021).
Nigrovic, P. A. et al. Anakinra as first-line disease-modifying therapy in systemic juvenile idiopathic arthritis: report of forty-six patients from an international multicenter series. Arthritis Rheum. 63, 545–555 (2011).
Ter Haar, N. M. et al. Treatment to target using recombinant interleukin-1 receptor antagonist as first-line monotherapy in new-onset systemic juvenile idiopathic arthritis: results from a five-year follow-up study. Arthritis Rheumatol. 71, 1163–1173 (2019).
Pardeo, M. et al. Early treatment and IL1RN single-nucleotide polymorphisms affect response to anakinra in systemic juvenile idiopathic arthritis. Arthritis Rheumatol. 73, 1053–1061 (2021).
Henderson, L. A. et al. Th17 reprogramming of T cells in systemic juvenile idiopathic arthritis. JCI Insight 5, https://doi.org/10.1172/jci.insight.132508 (2020).
Levescot, A. et al. IL-1β-driven osteoclastogenic Tregs accelerate bone erosion in arthritis. J. Clin. Invest. 131, https://doi.org/10.1172/JCI141008 (2021).
Alivernini, S. et al. Inclusion of synovial tissue-derived characteristics in a nomogram for the prediction of treatment response in treatment-naive rheumatoid arthritis patients. Arthritis Rheumatol. 73, 1601–1613 (2021).
Bergstra, S. A. et al. Earlier is better when treating rheumatoid arthritis: but can we detect a window of opportunity? RMD Open 6, https://doi.org/10.1136/rmdopen-2020-001242 (2020).
Goekoop-Ruiterman, Y. P. et al. Clinical and radiographic outcomes of four different treatment strategies in patients with early rheumatoid arthritis (the BeSt study): a randomized, controlled trial. Arthritis Rheum. 52, 3381–3390 (2005).
Wevers-de Boer, K. et al. Remission induction therapy with methotrexate and prednisone in patients with early rheumatoid and undifferentiated arthritis (the IMPROVED study). Ann. Rheum. Dis. 71, 1472–1477 (2012).
Ebrahimian, S. et al. Can treating rheumatoid arthritis with disease-modifying anti-rheumatic drugs at the window of opportunity with tight control strategy lead to long-term remission and medications free remission in real-world clinical practice? A cohort study. Clin. Rheumatol. 40, 4485–4491 (2021).
Krijbolder, D. I. et al. Intervention with methotrexate in patients with arthralgia at risk of rheumatoid arthritis to reduce the development of persistent arthritis and its disease burden (TREAT EARLIER): a randomised, double-blind, placebo-controlled, proof-of-concept trial. Lancet 400, 283–294 (2022).
Cope, A. P. et al. Abatacept in individuals at high risk of rheumatoid arthritis (APIPPRA): a randomised, double-blind, multicentre, parallel, placebo-controlled, phase 2b clinical trial. Lancet 403, 838–849 (2024).
Sugiyama, D. et al. Impact of smoking as a risk factor for developing rheumatoid arthritis: a meta-analysis of observational studies. Ann. Rheum. Dis. 69, 70–81 (2010).
Anderson, J. J., Wells, G., Verhoeven, A. C. & Felson, D. T. Factors predicting response to treatment in rheumatoid arthritis: the importance of disease duration. Arthritis Rheum. 43, 22–29 (2000).
Aletaha, D. et al. Effect of disease duration and prior disease-modifying antirheumatic drug use on treatment outcomes in patients with rheumatoid arthritis. Ann. Rheum. Dis. 78, 1609–1615 (2019).
Kerschbaumer, A. et al. Efficacy of synthetic and biological DMARDs: a systematic literature review informing the 2022 update of the EULAR recommendations for the management of rheumatoid arthritis. Ann. Rheum. Dis. 82, 95–106 (2023).
Chang, C. Y., Meyer, R. M. & Reiff, A. O. Impact of medication withdrawal method on flare-free survival in patients with juvenile idiopathic arthritis on combination therapy. Arthritis Care Res. 67, 658–666 (2015).
Guzman, J. et al. The risk and nature of flares in juvenile idiopathic arthritis: results from the ReACCh-Out cohort. Ann. Rheum. Dis. 75, 1092–1098 (2016).
Simonini, G. et al. Flares after withdrawal of biologic therapies in juvenile idiopathic arthritis: clinical and laboratory correlates of remission duration. Arthritis Care Res. 70, 1046–1051 (2018).
Curtis, J. R. et al. Etanercept or methotrexate withdrawal in rheumatoid arthritis patients in sustained remission. Arthritis Rheumatol. 73, 759–768 (2021).
Emery, P. et al. Adalimumab dose tapering in patients with rheumatoid arthritis who are in long-standing clinical remission: results of the phase IV PREDICTRA study. Ann. Rheum. Dis. 79, 1023–1030 (2020).
Ringold, S. et al. Disease recapture rates after medication discontinuation and flare in juvenile idiopathic arthritis: an observational study within the childhood arthritis and rheumatology research alliance registry. Arthritis Care Res. 75, 715–723 (2023).
Richmond, J. M. et al. Antibody blockade of IL-15 signaling has the potential to durably reverse vitiligo. Sci. Transl. Med. 10, https://doi.org/10.1126/scitranslmed.aam7710 (2018).
Hassert, M. et al. Regenerating murine CD8+ lung tissue resident memory T cells after targeted radiation exposure. J. Exp. Med. 221, https://doi.org/10.1084/jem.20231144 (2024).
Schenkel, J. M., Fraser, K. A. & Masopust, D. Cutting edge: resident memory CD8 T cells occupy frontline niches in secondary lymphoid organs. J. Immunol. 192, 2961–2964 (2014).
Beura, L. K. et al. T cells in nonlymphoid tissues give rise to lymph-node-resident memory T cells. Immunity 48, 327–338.e5 (2018).
Stolley, J. M. et al. Retrograde migration supplies resident memory T cells to lung-draining LN after influenza infection. J. Exp. Med. 217, https://doi.org/10.1084/jem.20192197 (2020).
Klicznik, M. M. et al. Human CD4+CD103+ cutaneous resident memory T cells are found in the circulation of healthy individuals. Sci. Immunol. 4, https://doi.org/10.1126/sciimmunol.aav8995 (2019).
Mijnheer, G. et al. Compartmentalization and persistence of dominant (regulatory) T cell clones indicates antigen skewing in juvenile idiopathic arthritis. Elife 12, https://doi.org/10.7554/eLife.79016 (2023).
Spreafico, R. et al. A circulating reservoir of pathogenic-like CD4+ T cells shares a genetic and phenotypic signature with the inflamed synovial micro-environment. Ann. Rheum. Dis. 75, 459–465 (2016).
Leijten, E. F. et al. Tissue-resident memory CD8+ T cells from skin differentiate psoriatic arthritis from psoriasis. Arthritis Rheumatol. 73, 1220–1232 (2021).
Kunnamo, I., Kallio, P., Pelkonen, P. & Viander, M. Serum-sickness-like disease is a common cause of acute arthritis in children. Acta Paediatr. Scand. 75, 964–969 (1986).
Lawley, T. J. et al. A prospective clinical and immunologic analysis of patients with serum sickness. N. Engl. J. Med. 311, 1407–1413 (1984).
Steere, A. C. et al. Treatment of Lyme arthritis. Arthritis Rheum. 37, 878–888 (1994).
Oen, K. et al. Early predictors of longterm outcome in patients with juvenile rheumatoid arthritis: subset-specific correlations. J. Rheumatol. 30, 585–593 (2003).
Selvaag, A. M., Aulie, H. A., Lilleby, V. & Flato, B. Disease progression into adulthood and predictors of long-term active disease in juvenile idiopathic arthritis. Ann. Rheum. Dis. 75, 190–195 (2016).
Lengl-Janssen, B., Strauss, A. F., Steere, A. C. & Kamradt, T. The T helper cell response in Lyme arthritis: differential recognition of Borrelia burgdorferi outer surface protein A in patients with treatment-resistant or treatment-responsive Lyme arthritis. J. Exp. Med. 180, 2069–2078 (1994).
Steere, A. C. et al. Antibiotic-refractory Lyme arthritis is associated with HLA−DR molecules that bind a Borrelia burgdorferi peptide. J. Exp. Med. 203, 961–971 (2006).
James, E. A. et al. Citrulline-specific Th1 cells are increased in rheumatoid arthritis and their frequency is influenced by disease duration and therapy. Arthritis Rheumatol. 66, 1712–1722 (2014).
Nigrovic, P. A. & White, P. H. Care of the adult with juvenile rheumatoid arthritis. Arthritis Rheum. 55, 208–216 (2006).
Acknowledgements
M.H.C. acknowledges related grant support from K08AR080992, the Rheumatology Research Foundation, the Arthritis National Research Foundation, Boston Children’s Hospital and the Arbuckle Family Foundation. R.C.F. acknowledges related grant support from R01AR075906 and P30AR079369. P.A.N. acknowledges related grant support from NIH/NIAMS grants R01AR073201, R01AR075906 and P30AR070253, the Rheumatology Research Foundation and the Arbuckle Family Foundation.
Author information
Authors and Affiliations
Contributions
All authors contributed equally to all aspects of the article.
Corresponding author
Ethics declarations
Competing interests
P.A.N. declares consulting relationships with Alkermes, Apollo, BMS, Exo Therapeutics, Fresh Tracks Therapeutics, Merck, Novartis, Pfizer, Qiagen and Sobi; equity in Edelweiss Immune Inc.; investigator-initiated research grants from BMS and Pfizer; and authorship and editorial income from UpToDate, the American Academy of Paediatrics and Arthritis & Rheumatology. M.H.C. and R.C.F. declare no competing interests.
Peer review
Peer review information
Nature Reviews Rheumatology thanks Femke Van Wijk, Stefano Alivernini and Sytske Anne Bergstra for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Cell spread model
-
New joints become inflamed when cells originating from an arthritic joint (such as resident memory T cells or fibroblasts) enter the circulation and deposit in previously unaffected joints.
- Founder model
-
New joints become inflamed when pathogenic cells seeded during an index event expand past a threshold that renders the joint symptomatic; joints cross this threshold at different times, depending on local factors that include the size of the initial inoculum.
- Irreversible steps
-
Durable changes that occur in tissues exposed to inflammation; such changes could include, for example, the accumulation of tissue-resident memory T cells, fibroblast priming and changes in synovial tissue macrophage populations.
- Joint accumulation model
-
A conceptual paradigm in which the course of inflammatory arthritis reflects two distinct forces: systemic drivers of arthritis, through which disease begins and extends to new joints, and durable local factors that promote disease recurrence and severity within an individual joint, mediating joint-specific memory.
- Joint-specific memory
-
The phenomenon that inflammatory arthritis is more likely to recur in joints that were previously affected by disease than in comparable joints that were previously spared.
- Parabiotic mice
-
A pair of mice in which the circulatory systems have been surgically connected, typically at the level of the skin, such that cells in the circulation equilibrate between the two animals whereas cells resident in tissue do not.
- Priming
-
The process through which activation of a cell, such as a synovial fibroblast, renders the cell hyper-responsive to subsequent stimuli.
- Systemic driver model
-
New joints become inflamed through the same processes that originally initiated arthritis, for example, the deposition of arthritogenic T cells from the circulation.
- Systemic drivers of arthritis
-
Processes outside of joints that have the potential to ignite synovitis; examples include autoantibodies and arthritogenic effector T cells.
- Trained immunity
-
The acquisition of enhanced responsiveness to previously encountered stimuli, typically through changes in the epigenome, conferring durable immune ‘memory’ in innate immune cells.
- Window of opportunity
-
A period during which intervention can induce a durable change in the natural history of a disease.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chang, M.H., Fuhlbrigge, R.C. & Nigrovic, P.A. Joint-specific memory, resident memory T cells and the rolling window of opportunity in arthritis. Nat Rev Rheumatol 20, 258–271 (2024). https://doi.org/10.1038/s41584-024-01107-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41584-024-01107-7