At its essence, cancer is unrestrained cell growth. Identifying the specific molecular mechanisms that regulate these uncontrolled growth pathways holds the key to stopping cancer and potentially even reversing its course. Now, a research team headed by scientists at the University of California (UC), San Diego School of Medicine and the Ludwig Institute for Cancer Research at UC San Diego has identified an enzyme involved in remodeling the plasma membrane of multiple cancer cell types that is critical to both survival of tumors and their uncontrolled growth.
“Cancers are characterized not only by major changes in their genomes but also by profound shifts in how they take up and utilize nutrients to propel rapid tumor growth,” explained senior study investigator Paul Mischel, MD, a professor in the UC San Diego School of Medicine. “How do these diverse aspects fit together, and can they be taken advantage of, for the benefit of patients?”
Findings from the new study were published recently in Cell Metabolism through an article entitled “Oncogene Amplification in Growth Factor Signaling Pathways Renders Cancers Dependent on Membrane Lipid Remodeling.”
In the current study, researchers identified an enzyme called LPCAT1, whose levels increase in cancer and which plays a key role in tumor growth by changing the phospholipid composition of the cancer cells’ plasma membrane, allowing amplified and mutated growth factor signals to spur tumor growth.
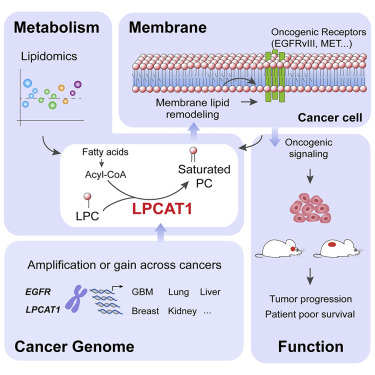
Interestingly, without LPCAT1, tumors cannot survive. When researchers genetically depleted LPCAT1 in multiple types of cancer in mice, including highly lethal glioblastomas (brain) and aggressive lung cancer, malignancies shrank dramatically, and survival times improved.
“Advances in DNA sequencing technologies have reshaped our understanding of the molecular basis of cancer, suggesting a new and more effective way of treating cancer patients,” the authors wrote. “However, to date, precision oncology has yet to benefit many patients, motivating a deeper search into understanding how genetic alterations in tumors change the way cancer cells behave, and potentially unlocking new ways to more effectively treat patients.
The authors concluded that the results demonstrate ”that LPCAT1 is an important enzyme that becomes dysregulated in cancer, linking common genetic alterations in tumors with changes in their metabolism to drive aggressive tumor growth. Moreover, these results also suggest that LPCAT1 may be a very compelling new drug target in a wide variety of cancer types.”